delivery within 72 Hours
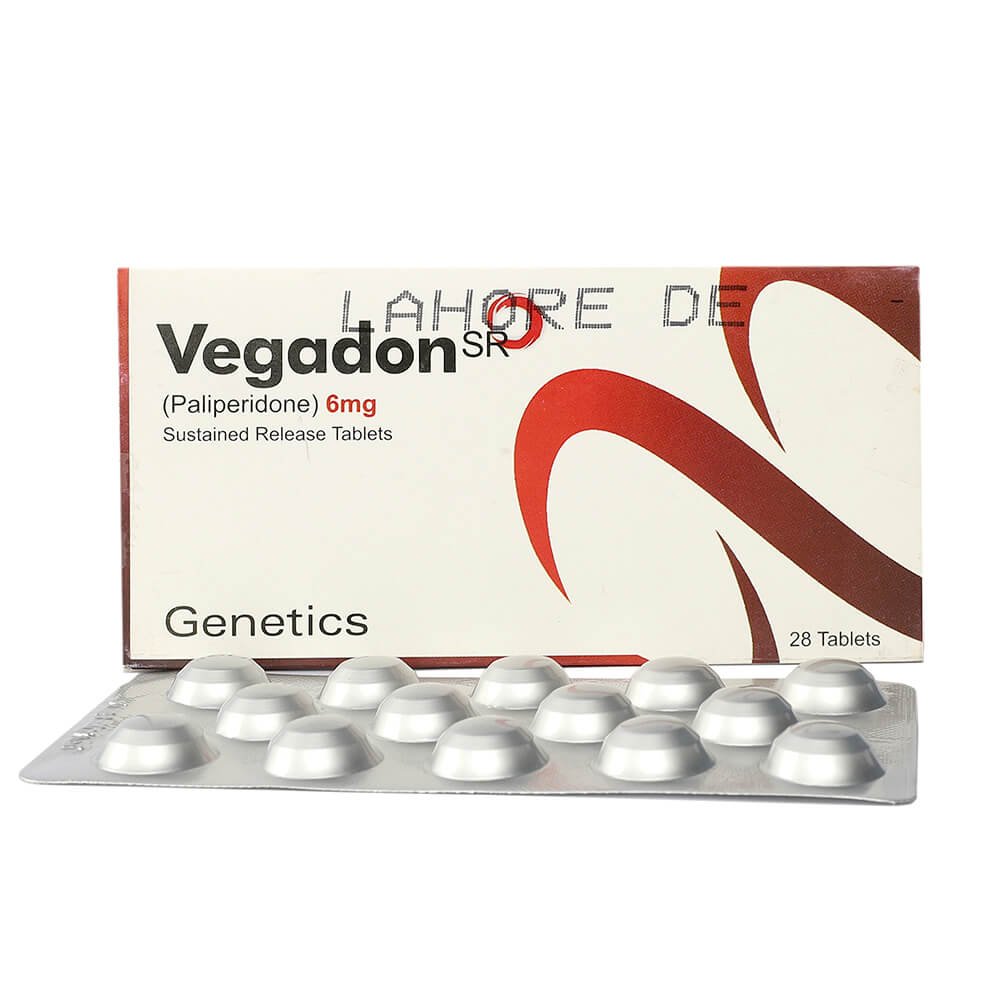
VEGADON SR 6MG TABLET
₨ 1,680
Generic: Paliperidone
Pack Size: 28
Product Form: TABLETS
Delivery within: 2 to 3 working days across Pakistan
Shipping & Delivery
-
Courier delivery
Our courier will deliver to the specified address
2-3 Days
From Rs 250
-
Free 15-Day returns

Black Friday Blowout!
Description
Vegadon SR 6mg tablet contains the active ingredient Paliperidone, which is an atypical antipsychotic medication. It is formulated as an extended-release tablet, designed to release the medication slowly into the body over an extended period. Paliperidone is used to treat various psychiatric disorders by affecting certain neurotransmitters in the brain.
Uses:
- Schizophrenia: Vegadon SR 6mg tablet is primarily prescribed for the treatment of schizophrenia, a severe mental disorder characterized by hallucinations, delusions, disorganized thinking, and emotional disturbances.
- Bipolar Disorder (Manic or Mixed Episodes): It is also used to manage bipolar disorder, specifically for controlling manic or mixed episodes.
Side Effects:
Paliperidone is generally well-tolerated, but some individuals may experience side effects. Common side effects of Vegadon SR 6mg tablet may include:
- Extrapyramidal Symptoms (EPS): These can include parkinsonian-like symptoms such as tremors, rigidity, and bradykinesia.
- Weight Gain: Some individuals may experience weight gain while taking Paliperidone.
- Drowsiness or Sedation: Vegadon SR 6mg tablet can cause drowsiness or sedation, particularly when starting the medication or with dosage adjustments.
- Dizziness: Paliperidone may lead to dizziness or a feeling of lightheadedness.
- Metabolic Changes: Vegadon SR 6mg tablet could potentially lead to changes in blood sugar levels, cholesterol levels, and weight.
- Orthostatic Hypotension: Some individuals may experience a drop in blood pressure upon standing, leading to dizziness.
- Sexual Dysfunction: Paliperidone may lead to sexual problems in some patients.
When Not to Use:
- Allergy: Vegadon SR 6mg tablet should not be used in individuals with a known hypersensitivity or allergy to Paliperidone or any other components of the medication.
- Dementia-Related Psychosis: Paliperidone is not approved for the treatment of dementia-related psychosis, as it may increase the risk of stroke and mortality in elderly patients with this condition.
- Severe CNS Depression: It should not be used in patients with severe central nervous system (CNS) depression or coma.
- Pregnancy and Breastfeeding: The safety of Vegadon SR 6mg tablet during pregnancy and breastfeeding has not been well-established, and its use in these situations should be carefully considered.
- Children and Adolescents: Paliperidone is not recommended for use in children and adolescents under the age of 18 years.
Important Note: Vegadon SR 6mg tablet is a prescription medication that should only be used under the guidance and supervision of a healthcare professional. Dosage adjustments and regular monitoring are essential, especially when using it for an extended period. Abrupt discontinuation should be avoided, as it may lead to the recurrence of symptoms.
Inform your doctor about any existing medical conditions or medications you may be taking before starting Vegadon SR 6mg tablet to avoid potential interactions or contraindications. If you experience any severe or persistent side effects, inform your healthcare provider promptly. Regular monitoring and follow-up with the healthcare provider are crucial during treatment with this medication.
Customer Reviews
You must be logged in to post a review.









Reviews
Clear filtersThere are no reviews yet.